α-Synuclein IMR Reagent
“MagQu” α-Synuclein IMR Reagent is designed for quantitative measurement of Tau protein concentration in human plasma by immunomagnetic reduction (IMR) assay. The reagent can be used with MagQu’s Magnetic Immunoassay Analyzer XacPro-S system.
This assay enables early-stage neurological disease research with ultra-high sensitivity and low interference.
Features
Quantify α-Synuclein in the sample easily, rapidly, and accurately
Magnetic Nanoparticle
Dextran layer
For Parkinson's disease research and for in-vitro diagnosis use
Specifications
Sample type: Human Plasma
Sample volume: 40 μl
Assay time: 5 hours (36 channels in XacPro-S)
Use application: In vitro diagnostic
Detection methods: ImmunoMagnetic Reduction (by analyzer XacPro-S with magnetic reagents)
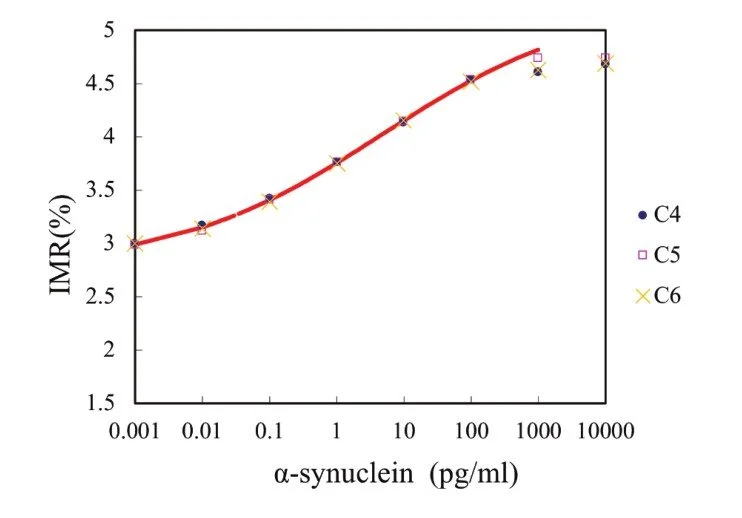
Sensitivity
Detection Range: 0.001 - 100 pg/ml
Low detection limit: 0.21 fg/ml
IMR standard curve of Tau Protein
Description
Intended Use
“MagQu” α-Synuclein IMR Reagent is used to quantitatively measure α-Synuclein in human fluid specimen, such as plasma, serum or CSF.
Use “MagQu” α-Synuclein IMR Reagent only with the XacPro-S System (MagQu Co., Ltd.).
Introduction
α-Synuclein (SNCA) is a presynaptic neuronal protein and is abundant in the human brain. α-Synuclein aggregates to form insoluble fibrils in pathological conditions characterized by Lewy bodies, such as Parkinson's disease (PD), dementia with Lewy bodies (DLB) and multiple system atrophy (MSA). These disorders are known as synucleinopathies. α-Synuclein is the primary structural component of Lewy body fibrils. Occasionally, Lewy bodies contain tau protein; however, α-Synuclein and tau constitute two distinctive subsets of filaments in the same inclusion bodies. α-Synuclein pathology is also found in both sporadic and familial cases with Alzheimer's disease.1, 2, 3
Principles of Test
“MagQu” α-Synuclein IMR reagent is designed for quantifying α-Synuclein protein by Immuno Magnetic Reduction (IMR). We conjugate antibody on the surface of around 50 nm-in-diameter Fe3O4 magnetic particles. When the antibodies on the surface bind with α-Synuclein protein, the magnetic particles form clusters. Therefore, the ac susceptibility (Xac) of magnetic particles would be reduced in the applied ac magnetic field. By measuring the reduction of Xac, we can quantify α-Synuclein protein in the sample easily and accurately.4, 5
Reagent Properties
Precision
The α-Synuclein samples were measured in dupli-
cate, twice per day over 20 days. The two measure-
ments on two sequent days are regarded as two
runs. Two different α-Synuclein concentrations
were used for the tests. The standard deviations of
repeatability and within-lab for various α-Synuclein
concentrations ware obtained.
Interference (Specificity)
Plasma can contain interfering substances such as hemoglo-
bin, bilirubin, or intra lipid because of common diseases, such
as hemolysis, jaundice or hypertriglyceridemia. Other bio-sub-
stances that exist naturally in plasma, such as uric acid,
rheumatoid factor, or albumin, are also interfering
substances. Other interfering substances include drugs or
chemicals in medicine that is used to treat inflammatory
diseases, viral and bacterial infections, cancers and cardio-
vascular disease. The level of α-Synuclein in each of these
pools was then determined and normalized to the level
without the respective substances.
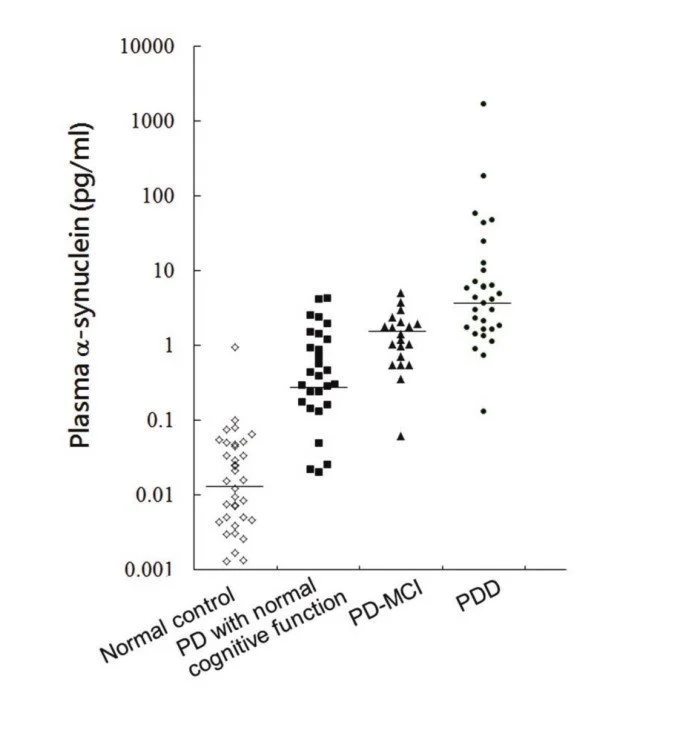
Expected Value
Fig 1: Plasma α-synuclein levels in PD patients with varying cognitive function. The plasma α-synuclein level was significantly higher in PD patients with more severe cognitive dysfunction than in PD patients with normal cognition (p<0.01). (refer to reference 1)
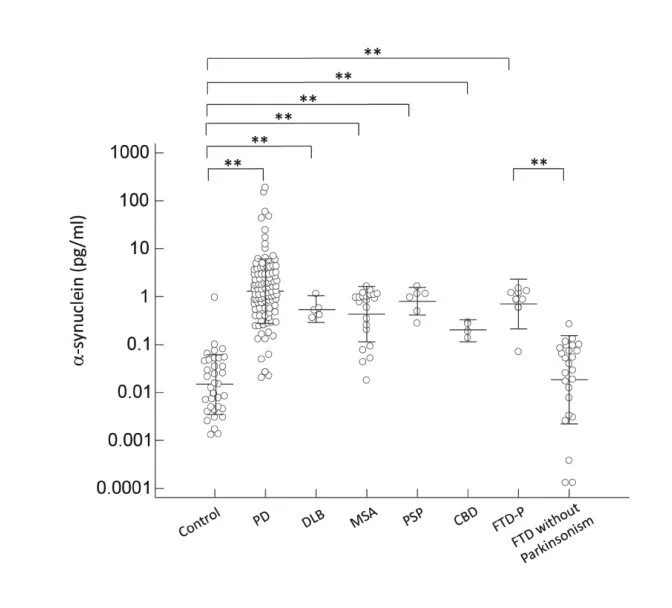
Fig 2: Plasma a-synuclein levels of normal controls and patients in different disease groups. The plasma a-synuclein level was significantly increased in patients with parkinsonism features compared to normal control participants and patients with FTD without parkinsonism (p < 0.001). (refer to reference 2)
Application References:
1. Lin, Chin-Hsien et al. “Plasma α-synuclein predicts cognitive decline in Parkinson's disease.” Journal of neurology, neurosurgery, and psychiatry. 2017; 88: 818
2. Lin, Chin-Hsien et al. “Plasma Biomarkers Differentiate Parkinson's Disease From Atypical Parkinsonism Syndromes.” Frontiers in aging neuroscience. 2018; 10